24 Sep Atomic Structure & The Changing Models of Atom
The following article will explain the timeline of the changing models of atom and the current model of the atomic structure. The students have started new specifications of OCR AS Chemistry A (H034),AQA AS Chemistry (7404),Edexcel AS Chemistry (8CH0) and CIE AS/A-level Chemistry (9701). All specifications require fundamental understanding of the changing models of atom and atomic structure. This article will be followed by atomic structure worksheet with video explanation of the answers. The ‘atomic structure worksheet’ will follow the new format of exams and contain multiple choice questions, short answer questions and extended answer questions on the changing models of atom and the atomic structure.
A-level Chemistry Specifications
This article will cover the following topics from A-level chemistry specifications:
OCR AS Chemistry
- The changing models of atomic structure over time and the use of evidence to accept or reject particular models.
- Atomic structure in terms of the numbers of protons, neutrons and electrons for atoms and ions, given the atomic number, mass number and any ionic charge.
AQA AS Chemistry
- Appreciate how the understanding of atom has evolved over time.
- An atomic structure in terms of a nucleus containing protons and neutrons surrounded by electrons.
Edexcel AS Chemistry
- Students should consider how models of atomic structure have developed over time, as new evidence has become available.
- Know the structure of an atom in terms of electrons, protons and neutrons.
CIE AS Chemistry
- Identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses.
- Describe the distribution of mass and charges within an atom.
- Deduce the numbers of protons, neutrons and electrons present in both atoms and ions given proton and nucleon numbers.
- Describe the contribution of protons and neutrons to atomic nuclei in terms of the proton number and nucleon number (ii) distinguish between isotopes on the basis of different numbers of neutrons present (iii) recognise and use the symbolism yxA for isotopes, where x is the nucleon number and y is the proton number.
Topic 1: The changing models of atomic structure
This topic will highlight the changing models of atomic structure and how sequence of experiments, observations, analysis and conclusions led to the modern day model of atom. The content of the article will be restricted to AS/A-level students.
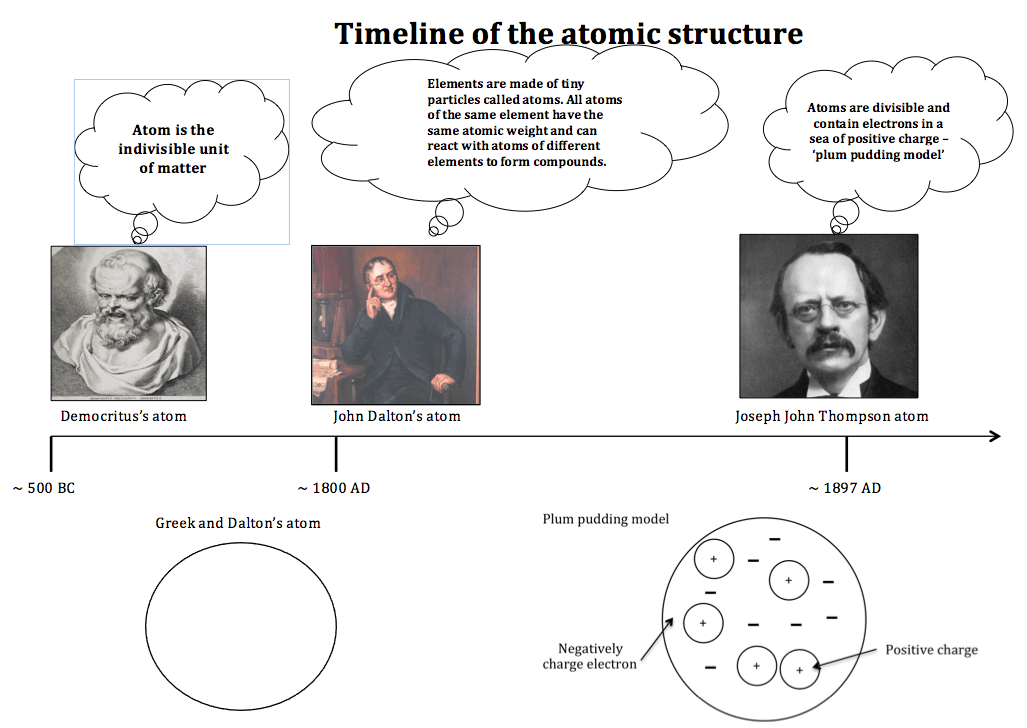
Democritus model of atom ~ 400 BC
Fig. 1. The Democritus concept of atom. If you keep cutting a metal eventually a point is reached where cutting the metal any further would not be possible. The smallest unit of metal is known as atom.
The concept of atom evolved from the understanding of Greek philosopher Democritus. The understanding was merely a philosophical & theological reasoning rather than based on hard evidence. He understood matter as being made up of tiny particles that were indivisible and called such particles atoms. In his view, there were many different types of atoms, which differ in shapes, sizes and nature of connectivities between them. For example, iron was made up of solid atoms with hooks locking atoms to each other, salt was made up of pointed and sharp atoms because of their taste and the atoms present in air were thought to be lightweight and swirling around in space.
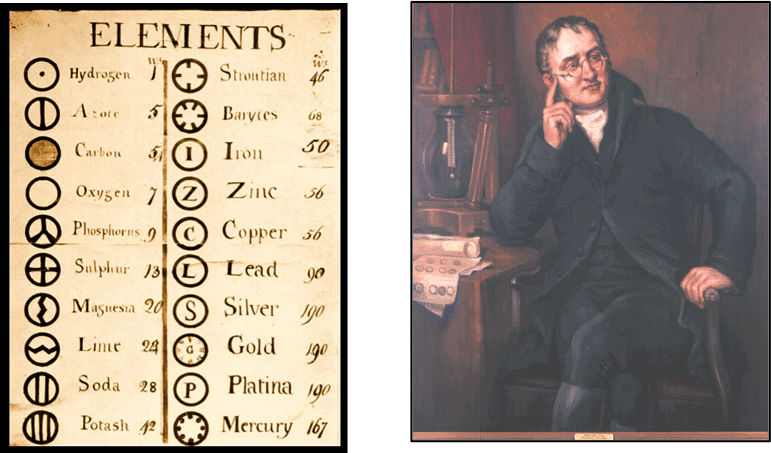
John Dalton’s atom ~1800
Fig. 2. John Dalton used his own symbols to represent atoms and also developed the first table of atomic masses in New system of Chemical Philosophy (1808).
John Dalton was the founder of the atomic theory. The main points of his atomic theory were:
- Elements are made up of tiny particles called atoms.
- Atoms cannot be divided, altered or destroyed by chemical means.
- Atoms of an element are all the same in terms of shape and size and atoms of different elements differ in size and shape.
- Elements are characterised by mass of their atoms so all the atoms of an element have identical weight. Different elements have atoms of different atomic weight.
- Atoms of different elements can combine together to form chemical compounds.
- When elements undergo chemical reactions, their atoms combine in simple, whole number ratios or more than one simple whole number ratios. Dalton noted that tin can combine with either one or two oxygen atoms which was in line with the percentages of masses observed for tin oxides. Similarly he used the same principle to explain that the different ratios of nitrogen to oxygen in various nitrogen oxides were simple multiples of each other.
However, John Dalton wrongly postulated that when atoms combine only in one ratio, it must be assumed to be binary (in two) one. He got formula of water as HO and ammonia as NH that were both incorrect as he got the atomic weights of oxygen and nitrogen incorrect. A lot of his own experimental evidence didn’t coincide with his theory of atomic structure and was at odds with the scientific community.
Joseph John Thompson’s model of atom ~ 1897
Until 1897 atoms were thought to be the smallest possible division of matter. However, J.J. Thompson discovered ‘electron’ through series of experiments on cathode rays, which disproved the notion that atoms were indivisible as proposed by ancient Greeks and Dalton. This led to the discovery of sub-atomic particles, which are considered to be the fundamental units of the modern day atomic structure.
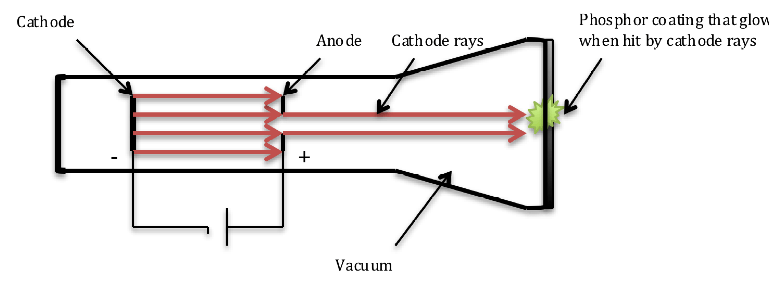
Fig. 3. Design of cathode ray tube (CRT) used by J.J Thompson to discover the nature of cathode rays.
Before we look into how J.J. Thompson discovered electron, we need to understand how cathode ray tube works. A cathode ray tube (CRT) is a sealed glass container in which a cathode (negatively charged) and anode (positively charged) are separated by a vacuum as shown in fig. 3. When a voltage is applied across the electrodes, cathode rays are generated from the cathode and are attracted towards the anode. A small hole in the anode allows some electrons to pass through creating a beam of cathode rays producing a glowing patch when they strike the phosphorous-coated glass at the opposite end of the tube or register an electric current when the end is attached to an electrometer. Earlier physicists had discovered cathode rays as yet unknown entity that registered an electric charge on an electrometer. Scientists debated whether cathode rays were immaterial like light or particles of matter. Thompson carried out a series of experiments using cathode rays generated in CRT and was able to demonstrate the following properties of the cathode rays:
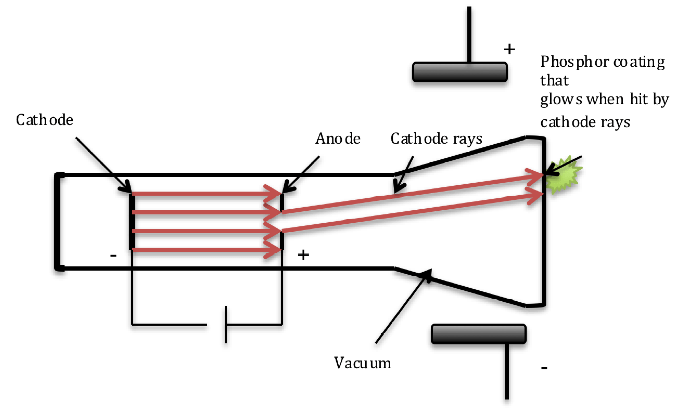
- Cathode rays were negatively charged particles. Thompson introduced metal plates on either side of the CRT to bend the cathode rays emerging from the hole present in the anode. He observed that cathode rays were deflected towards the positive electrode of the electric field indicating that cathode rays were negatively charged (Fig.4).
Fig. 4. The deflection of cathode rays by electric field.
- The particles were deflected by magnetic as well as electrical field.
- The mass of the particles was very, very small. The particles were about 2000 times lighter than the hydrogen ions.
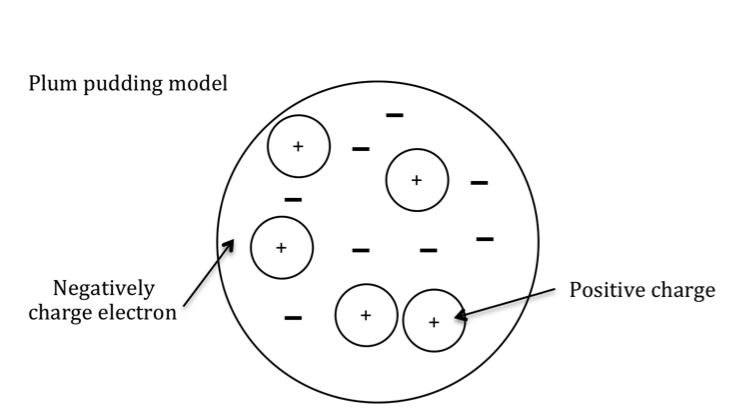
J.J.Thompson named the particle as ‘corpuscle’ but the name was later changed to ‘electron’. Since the electrons originated from within the electrodes, they led Thompson to conclude that atoms were divisible and that electrons (corpuscles) were the building blocks of the atom. He proposed that electrons were moving randomly in a sea of uniformly distributed positive charge to account for the neutral charge of the atom as plums in a plum pudding. This model is called the ‘plum pudding model’ of atomic structure (fig.5).
Fig.5. The plum pudding model of the atomic structure.
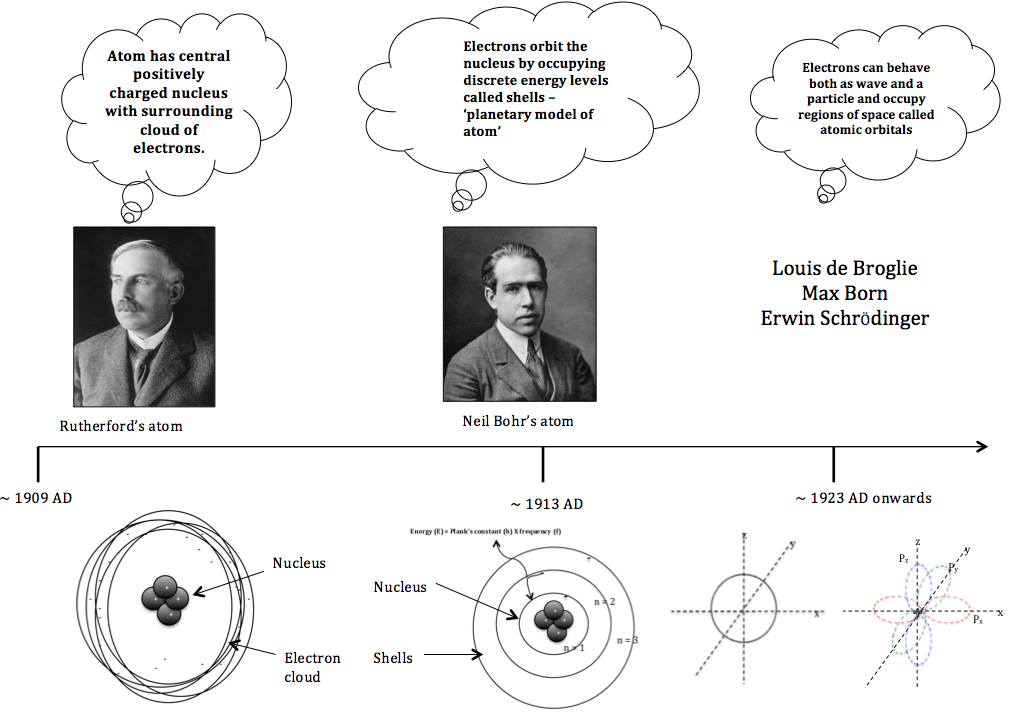
Ernest Rutherford model of atomic structure ~ 1909
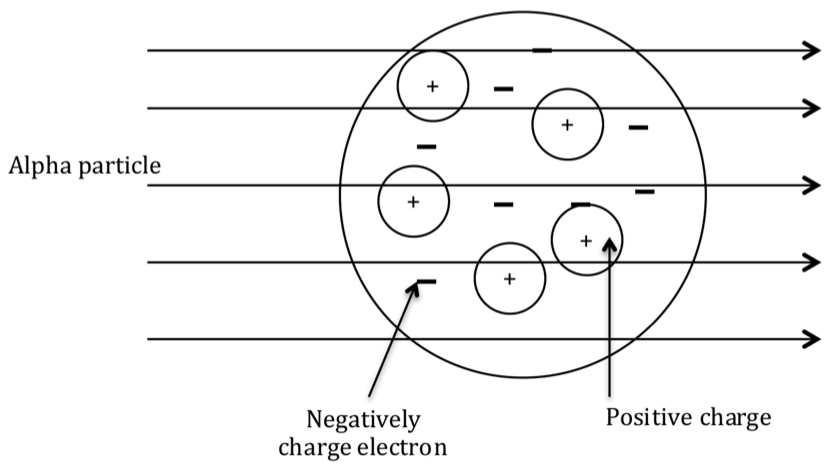
J.J. Thompsons plum-pudding model was disproved by the experiment carried out by Hans Geiger and Ernest Marsden (Geiger-Marsden experiment) working at the behest of Ernest Rutherford. This led to the development of the nuclear model of atomic structure. In Geiger-Marsden experiment (a.k.a gold-leaf experiment), alpha particles were fired at thin sheet of gold foil and the deflection of the particles was measured once they crossed the gold foil. Based on the Thompson’s plum pudding model of atom (small mass of electrons & low concentration of positive charge), the scientists expected that the alpha particles to pass through the gold foil without any deflection. They made following observations:
Expected results from the plum pudding model of Thompson
alpha particles passing through the atom with negligible deflection
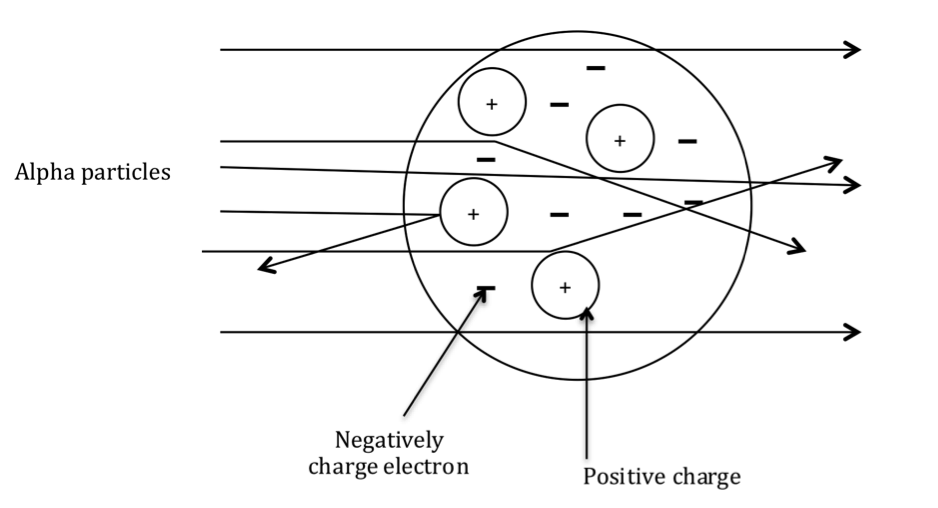
Actual results from the gold-leaf experiment:
A small proportion of particles were deflected by the dense positive charge of the nucleus.
1. Majority of the particles were not deflected at all.
2. A small percentage of particles were deflected through large angles.
3. A very small fraction of particles were deflected back to the source.
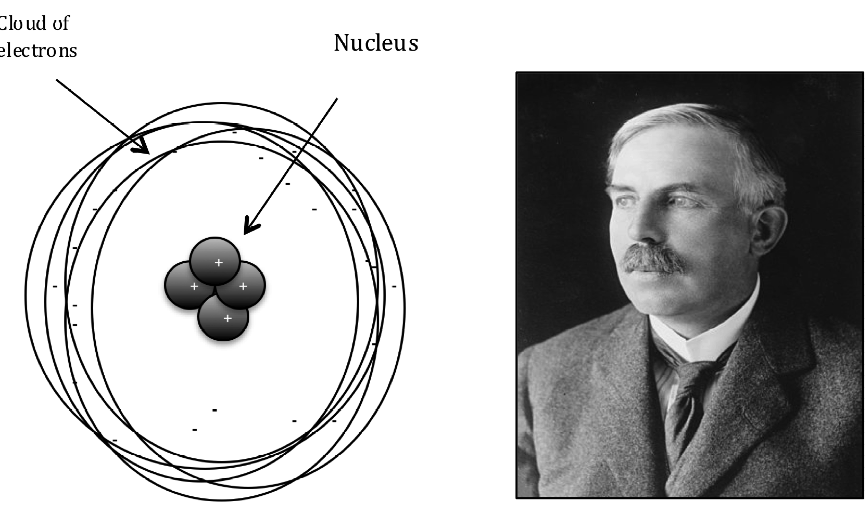
Rutherford concluded that the positive charge of the atom must be concentrated in a very small volume to produce electric field of sufficient strength to repel the alpha particles. Rutherford proposed the following model of nuclear atom in 1911 (fig.6).
1. The positive charge of an atom is concentrated in the nucleus, which was assumed to be present in the center.
2. Clouds of electrons orbit the nucleus.
3. Most of the atom’s volume lies between tiny nucleus and cloud of electrons.
4. There is no net charge on the atom as positive and negative charges cancel out each other.
Fig. 6. The proposed structure of the nuclear atom by Ernest Rutherford.
Henry Moseley’s contribution to the atomic structure ~ 1913
Before Mosley, the atomic number of elements was an arbitrary number assigned by scientists to order the elements in the periodic table. Moseley used a technique called X-ray spectroscopy to establish a mathematical relationship between the atomic number of the elements and the wavelength of the X-rays. Moseley’s work redefined atomic numbers into a value that can be used to order the elements in the periodic table. His work supported the concept of atomic structure put forward by Ernest Rutherford, which had compact positively charged nucleus surrounded by orbiting cloud of electrons. Moseley understood atomic number to be the exact number of positive charges (later found by Ernest Rutherford to be protons) in the nucleus of an atom.
Bohr’s model of atomic structure ~ 1913
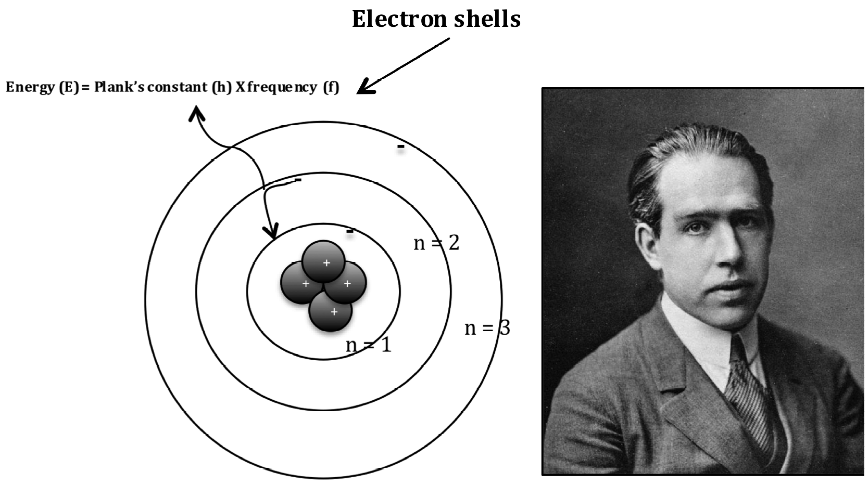
In 1913 Neils Bohr proposed the Bohr model of atom that incorporated the idea of quantum physics postulated by Albert Einstein and Max Planck that light energy is emitted or absorbed in discrete amounts known as quanta. His model allows electrons to follow certain paths (shells) around the nucleus with discrete energy. Under this model the electrons in shells could not loose energy and can only jump from one energy level to another. When this happened light was emitted or absorbed at a frequency proportional to the energy difference between shells (Fig.7). Bohr’s model of atomic structure helped explain some periodic properties such as spectral lines seen in emission spectra and the energy of electrons at different distances from the nucleus.
Fig. 7. The planetary model of Neils Bohr’s atomic structure.
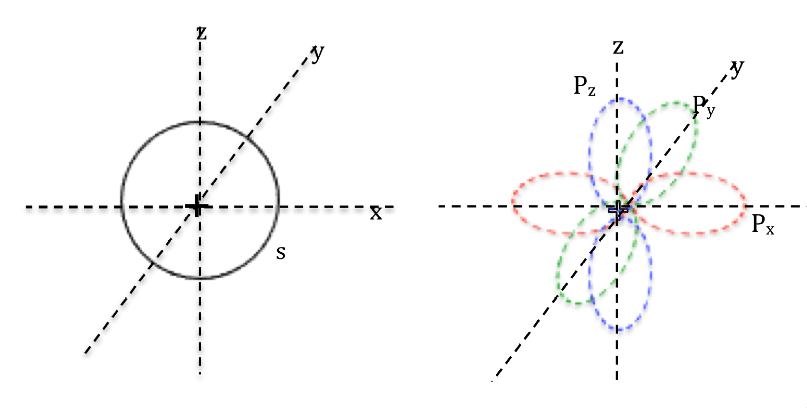
Quantum model of atomic structure ~ 1923
In 1924, Louis de Broglie proposed that the behaviour of electrons could be explained both in terms of wave and particle. In 1926 Erwin Schrödinger described electron as a wave particle in the form of Schrödinger equation. The equation was difficult to visualise and Max Born reconciled the opposing ideas of describing electron as wave and particle by introducing the theory of particle-wave duality. He proposed that Schrödinger equation describes the position of electrons in an atom in terms of probabilities. This invalidated the concept of Bohr’s Model of atom, which had clear defined paths of electrons in circular orbits. Rather the position of electrons in an atom is now defined in terms of probabilities. Based on this, the region with the highest probability of finding an electron around the nucleus is called the atomic orbital.
Quantum mechanics model
Electrons are found in regions of space (orbitals) whose shape is described by mathematical equations
Discovery of nuclear particles ~ 1917 – 1935
In 1918, Ernest Rutherford discovered protons from his work on bombarding nitrogen gas with alpha particles and observing the hydrogen nuclei being emitted from the gas. Further experimentation led him to conclude that the mass of the nucleus was greater than the protons it contained. This was attributed to the unknown neutral particles present in the nucleus, which he called neutrons. This was validated by the experiments of James Chadwick in 1932, who bombarded high-energy α-particles to beryllium and observed the mysterious highly penetrating and electrically neutral radiation. He further demonstrated that the neutral radiation had mass similar to proton and hence cannot be weight-less gamma rays. James Chadwick had discovered Rutherford’s neutrons.
Summary: Timeline of the atomic structure
Topic 2: Atomic Structure
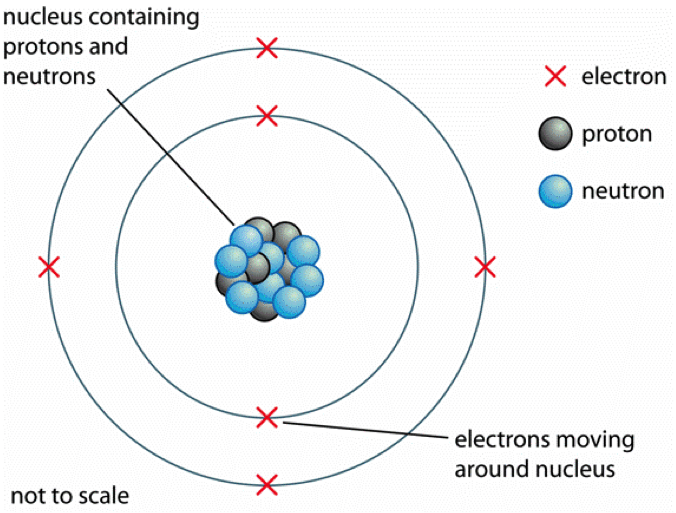
Current model of atomic structure
The current model of the structure of an atom is as follows:
- The nucleus contains protons and neutrons, which are located at the center of the atom.
- Electrons orbit the nucleus in shells, which contain the set of orbitals.
- The nucleus is small, dense mass at the centre of the atom.
- Most of the atom is empty space between the nucleus and the electron shells.
Fig. 1 The current model of the atomic structure.
Table 1 shows the relative mass and charge of sub-atomic particles.
- Protons are positively charged and electrons are negatively charged while neutrons have no charge.
- An atom has same number of protons and electrons so the atom is electrically neutral.
- A proton has same mass as a neutron.
Table 1 shows charges and masses of sub-atomic particles.
| Particle | Relative mass | Relative charge |
| Proton | 1.0 | 1+ |
| Neutron | 1.0 | 0 |
| Electron | 1/2000 | 1- |
Isotopes
- Isotopes are atoms of the same element with the same number of protons, but different numbers of neutrons.
- Isotopes have similar chemical properties (react in a similar way) because they have the same electronic structure (number of electrons in the outer shells).
- Isotopes may have slightly varying physical properties because of different masses (e.g. different melting point and boiling points).
- Elements do not usually contain all identical atoms but are made up of a mixture of isotopes. The natural abundances may vary in different sources.
Symbol notation of elements in the periodic table
Carbon and hydrogen isotopes
Table 2 – Atomic structures of hydrogen and carbon isotopes.
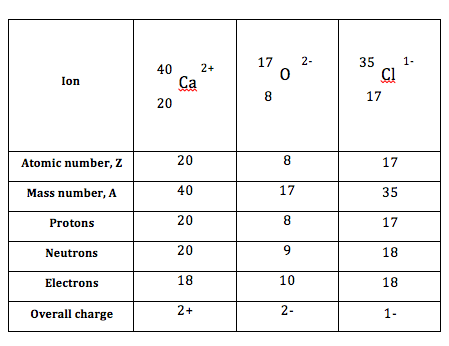
Atomic structure of ions
Atoms form ions by gaining and loosing electrons. The ions are charged because the positively charged protons and negatively charged electrons do not cancel each other. The electrons are lost to form positively charged ions while electrons are gained to form negatively charged ions.
Table 4 show examples of the atomic structure of ions.
This completes the AS level topic on the changing models of atom and atomic structure.
Atomic Structure Worksheet
DOWNLOAD ATOMIC STRUCTURE WORKSHEET
SCROLL DOWN FOR VIDEO SOLUTION TO THE ATOMIC STRUCTURE WORKSHEET
Section 1: Multiple Choice Questions
Q1. The isotope cobalt-60 (60Co) is used to destroy cancer cells in the human body.
Which statements about an atom of cobalt-60 are correct?
1. It contains 33 neutrons.
2. Its nucleus has a relative charge of 27+.
3. It has a different number of neutrons from the atoms of other isotopes of cobalt.
(A) 1, 2 & 3 are correct.
(B) 1 & 2 are correct.
(C) 2 & 3 are correct.
(D) 1 only is correct.
Q2. In which species are the numbers of electrons and neutrons equal?
A 9Be
B 19F
C 23Na+
D 18O2–
Q3. A radioactive isotope of thallium, 20181Tl, is used to assess damage in heart muscles after a heart attack.
Which statement about 201 81Tl is correct?
A This isotope has a nucleon number of 120.
B The number of electrons in one atom of this isotope is 81.
C The number of neutrons in one atom of this isotope is 201.
D 201 82X is an isotope of 201 81Tl.
Q4. John Dalton’s atomic structure theory, published in 1808, contained four predictions about atoms. Which of his predictions is still considered to be correct?
A Atoms are very small in size.
B No atom can be split into simpler parts.
C All the atoms of a particular element have the same mass.
D All the atoms of one element are different in mass from all the atoms of other elements.
Q5. The technetium–99 isotope (99Tc) is radioactive and has been found in lobsters and seaweed adjacent to nuclear fuel reprocessing plants.
Which statements are correct about an atom of 99Tc?
1 It has 13 more neutrons than protons.
2 It has 43 protons.
3 It has 99 nucleons.
(A) 1, 2 & 3 are correct.
(B) 1 & 2 are correct.
(C) 2 & 3 are correct.
(D) 1 only is correct.
Q6. Skin cancer can be treated using a radioactive isotope of phosphorus, 32P. A compound containing the phosphide ion 3215P3−, wrapped in a plastic sheet, is strapped to the affected area.
What is the composition of the phosphide ion, 3215P3− ?
Q7. One of the most important chemical species responsible for the removal of ozone from the stratosphere is a free radical of chlorine, 35Cl•.
What is the atomic structure of 35Cl• ?
Q8. Unnilpentium is an artificial element. One of its isotopes is 262105Unp. Which of the following statements is correct?
A 262105Unp has a nucleon number of 105.
B The atom 260X is an isotope of 262105Unp.
C There are 262 neutrons in 262105Unp.
D The proton number of 262105Unp is 262.
Q9. Use of the Data Booklet is relevant to this question.
It is now thought that where an element exists as several isotopes, the stable ones usually contain a ‘magic number’ of neutrons. One of these magic numbers is 126.
Which isotope is unstable?
A 209Bi B 208 Pb C 210Po D 208 Tl
Q10. Which ion has more electrons than protons and more protons than neutrons?
[H = 11H; D = 2 1H; O = 168 O]
A D– B H3O+ C OD– D OH–
Q11. Use of the Data Booklet is relevant to this question.
Hard water contains calcium ions and hydrogencarbonate ions arising from dissolved calcium hydrogencarbonate, Ca(HCO3)2.
How many electrons are present in the hydrogencarbonate anion?
A 30 B 31 C 32 D 33
Q12. The nucleus of a radon atom contains
A 222 protons and 86 neutrons B 86 protons and 136 neutrons
C 86 protons and 222 neutrons D 86 protons, 136 neutrons and 86 electrons
Q13. In which pair do the species contain the same number of neutrons?
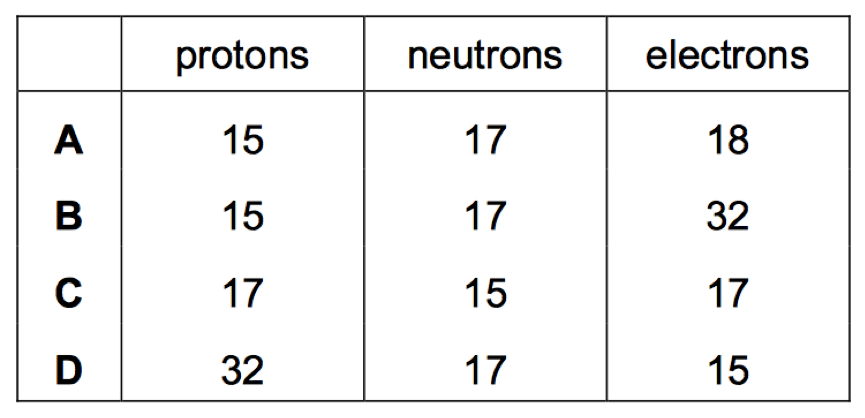
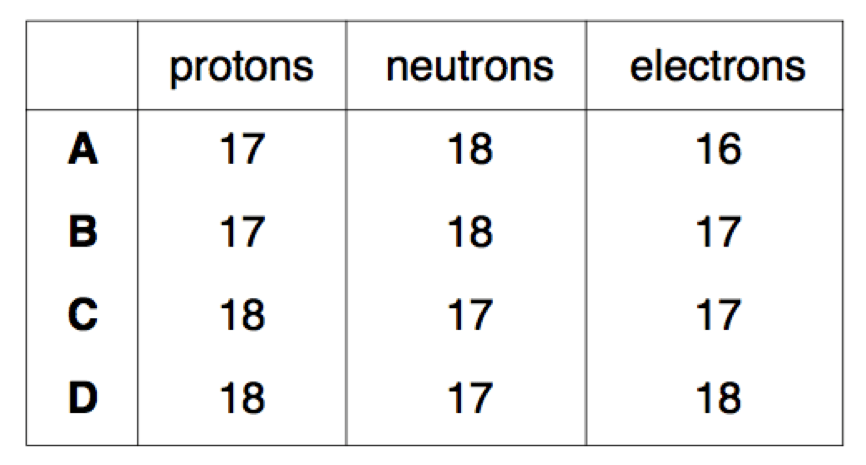
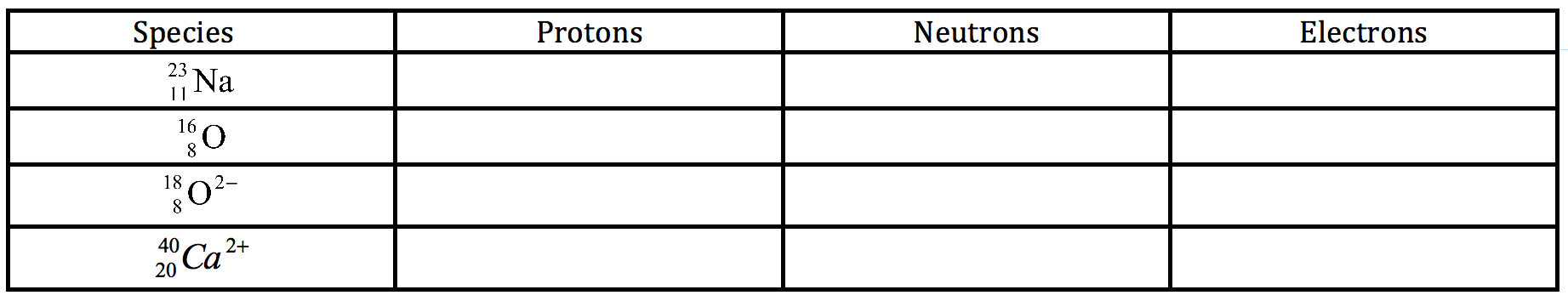
Q14. Complete the table below by giving the numbers of protons, neutrons and electron [4]
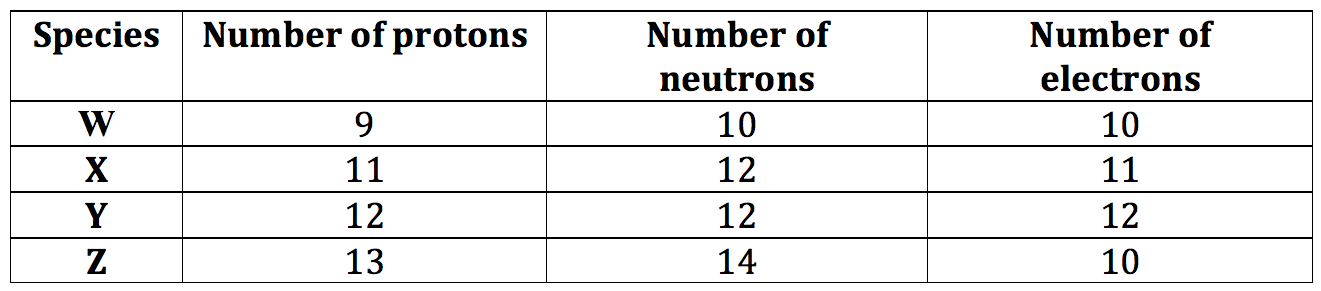
Q15. Consider the composition of the species W, X, Y and Z below. Which species is a positive ion?
A W B X C Y D Z
Q16. Chlorine occurs as two natural isotopes, Chlorine-35 and Chlorine-37. Which one of the following statements concerning chlorine atoms is FALSE.
A All chlorine atoms have the same nuclear charge.
B The nuclei have diameters which are approximately 1/10 000 of the diameter of chlorine atoms.
C Some naturally occurring chlorine nuclei contain 18 neutrons.
D Some naturally occurring chlorine nuclei contain 20 electrons.
Q17. Isotopes are elements with
A the same atomic number and the same number of neutrons
B the same mass number but a different number of neutrons
C the same atomic number but a different number of neutrons
D different atomic and mass numbers but the same number of neutrons
Q18. Which of the following particles contains more electrons than neutrons
A I only B II only C I and II only D II and III only
Q.19 Which species has 54 electrons and 52 protons?
(A) 12852Te2−
(B) 132 54Xe2+
(C) 132 54Xe2−
(D) 128 52Te2+
Q20. Which of the following represents a pair of isotopes?
A 146C and 147N
B 3216S and 3216S2–
C O2 and O3
D 20682Pb and 20882Pb
Q21. An isotope of an element, atomic number z, has mass number 2z + 4. How many neutrons are in the nucleus of the element?
A z+ 4
B z+ 2
C z
D 4
Q22. The nucleus of a 23 11Na atom contains
A 11 protons and 12 neutrons.
B 11 protons and 12 electrons.
C 23 protons and 11 neutrons.
D 23 protons and 11 electrons.
Q23. The isotopes of magnesium, 2412Mg and 2512Mg, both form ions with charge 2+. Which of the following statements about these ions is true?
A Both ions have electronic configuration 1s2 2s2 2p6 3s2.
B 25Mg2+ has more protons than 24Mg2+.
C The ions have the same number of electrons but different numbers of neutrons.
D The ions have the same number of neutrons but different numbers of protons.
Q24. Which of the following does not have exactly 10 electrons?
A An ion of fluorine, F–
B A molecule of methane, CH4
C A molecule of nitrogen, N2
D An ion of sodium, Na+
Q25. Which of the following statements about 2512Mg is true?
(A) It has the same number of neutrons and protons.
(B) It can be oxidized to form an Mg2+
(C) It is the most common isotope of magnesium.
(D) It has higher ionization energy than 2412Mg.
Q26.
Section 2: Atomic structure & the changing models of atom (Short Answer Questions)
Q1. Sir James Jeans, who was a great populariser of science, once described atomic structure of carbon as being like six bees buzzing around a space the size of a football stadium.
(a) (i) Suggest what were represented by the six bees in this description.
……………………………………………………………………………………………………………………………….[1]
(ii) Explain (in terms of an atom of carbon) what stopped the bees from flying away from the space of the football stadium.
…………………………………………………………………………………………………………………………………..
……………………………………………………………………………………………………………………………….[1]
(iii) What is missing from Jeans’ description when applied to an atom of carbon?
…………………………………………………………………………………………………………………………………..
……………………………………………………………………………………………………………………………….[1]
Q2. (a) Define an isotope in terms of its sub-atomic particles.
………………………………………………………………………………………………………………………………….
………………………………………………………………………………………………………………………………..[2]
Q3. Iron and cobalt are adjacent elements in the Periodic Table. Iron has three main naturally occurring isotopes, cobalt has one.
(a) Explain the meaning of the term isotope ? …………………………………………………………………………………………………………………………………………………………………………………………………….. ……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………..[2]
(b) The most common isotope of iron is 56Fe; the only naturally occurring isotope of cobalt is 59Co. Use the Data Booklet to complete the table below to show the atomic structure of 56Fe and of 59Co.
Q4. In the 19th and 20th centuries, scientists established the atomic theory and showed that three sub-atomic particles, electron, neutron and proton, exist. The masses and charges of these three particles were subsequently determined.
(i) Define the term proton number.
…………………………………………………………………………………………………………………. …………………………………………………………………………………………………………………[1]
(ii) Why is the proton number of an atom of an element usually different from the nucleon number of an atom of the element?
………………………………………………………………………………………………………………….
…………………………………………………………………………………………………………………[1]
(iii) Protons and neutrons have been used in nuclear reactions which result in the formation of artificial elements. In such processes, protons or neutrons are accelerated to high speeds and then fired like ‘bullets’ at the nucleus of an atom of an element.
Suggest why neutrons are more effective than protons as ‘nuclear bullets’.
………………………………………………………………………………………………………………………
…………………………………………………………………………………………………………………..[2]
(iv) In some cases, when neutrons are fired at atoms of an element, the neutrons become part of the nucleus of those atoms.
What effect does the presence of an extra neutron have on the chemical properties of the new atoms formed? Explain your answer.
………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………
…………………………………………………………………………………………………………………..[2]
Q5. The relative atomic masses of the elements are based upon the 12C scale.
(a) What is the composition of the 12C nucleus?
……………………………………………………………………………………..[1]
(b) Carbon also exists as the 14C isotope. How does the composition of the 14C isotope differ from that of the 12C isotope?
…………………………………………………………………………………………………………………… [1]
Q6. Hydrogen has three isotopes, 1H, known as protium, 2H, deuterium, and 3H, tritium.
(a) In terms of sub-atomic particles, give the similarities and differences between atoms of these three isotopes of hydrogen.
………………………………………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………………………………………………………
………………………………………………………………………………………………………………………………………………………………………………….[3]
Q7. (a) Give the relative charge and relative mass of an electron.
Relative charge
.…………………………………………………………………………………….[1]
Relative mass
…………………………………………………………………………………….[1].
(b) Isotopes of chromium include 54Cr and 52Cr
(i) Give the number of protons present in an atom of 54Cr
……………………………………………………………………………………..[1]
(ii) Deduce the number of neutrons present in an atom of 52Cr
……………………………………………………………………………………..[1]
Q8. (a) Complete the following table. [2]
| Relative mass | Relative charge | |
| Proton | ||
| Electron |
(b) An atom of element Q contains the same number of neutrons as are found in an atom of 27Al. An atom of Q also contains 14 protons.
(i) Give the number of protons in an atom of 27Al.
………..………………………………………………………………[1]
(ii) Deduce the symbol, including mass number and atomic number, for this atom of element Q.
………..………………………………………………………………………………………………………………………[1]
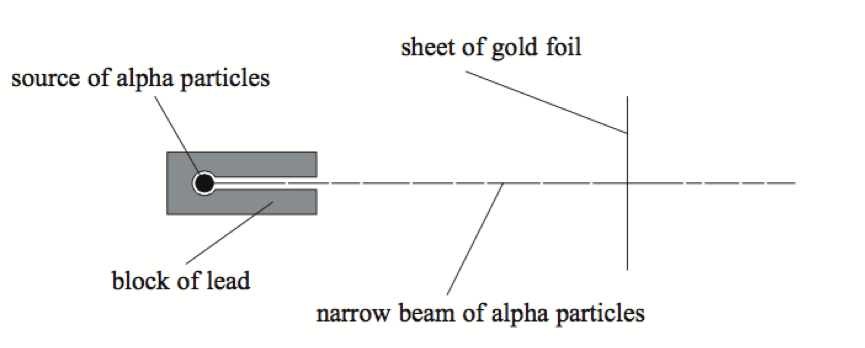
Q10. Geiger and Marsden studied the structure of atoms. The diagram shows part of the equipment, which they used.
(i) The block of lead helped to shield the scientists from radiation. State another purpose of the block of lead. ……………………………………………………………………………………………………………………………………………………………………………………………………………………….. (1)
(ii) Most of the alpha particles went straight through the gold foil. State another purpose of the block of lead. ………………………………………………………………………………………………………………………………………………………………………………………………………………………..(1)
(iii) Some of the alpha particles were deflected. What explanation did the scientists suggest for the deflection? ………………………………………………………………………………………………………………………………………………………………………….. …………………………………………… (2)
(iv) Only a small proportion of the alpha particles deflected through a large angle. What explanation did the scientists suggest for the proportion being small? …………………………………………………………………………………………………………………………………………………………………………………………………………………………(1)
(v) Some alpha particles were deflected less than others. What two reasons did the scientists suggest for this? …………………………………………………………. ………………………………………………………………………………………………………………………………………………………………………………………………………………………… (2)
(vi) The alpha particles were detected when they hit a zinc sulphide screen. How did the scientists know that an alpha particle had hit the screen?
……………………………………………………………………………………………………………….. …………………………………………………………………………………………………………[1]
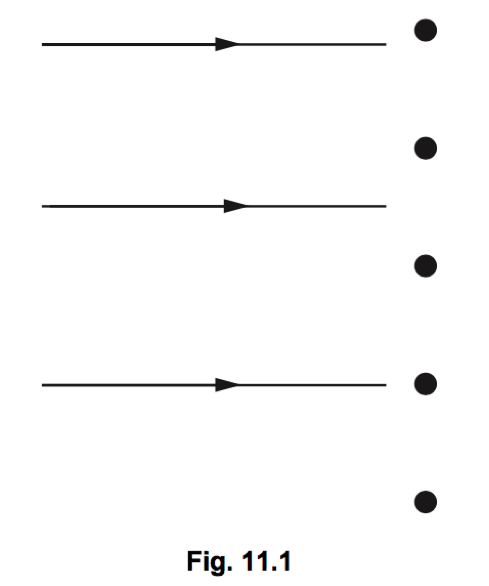
Q11. In Geiger and Marsden’s α-particle scattering experiment, α-particles were directed at a very thin gold foil. Fig. 11 shows five of the nuclei of the atoms in one layer in the gold foil. Also shown are the paths of three α-particles directed at the foil.
(a) On Fig.11.1, complete the paths of the three α-particles. [3]
(b) (i) State the result of the experiment that shows that an atom consists of a very tiny, charged core, containing almost all the mass of the atom. [1]
(ii) State the sign of the charge on this core. ……………………………………………………………………………………………………………………………………………….. [1]
(iii) State what occupies the space between these charged cores. [1]
(c) The nuclide notation for an α-particle is 42 α. State the number of protons and neutrons in an α-particle [2]
protons = ——————————————————————————————————————-
Neutrons= —————————————————————————————————————–
Q12. Use the following information to help you answer the questions.
The gold foil experiment
Scientists used to think that electrons were spread out through a positively charged atom. They called this the ‘plum pudding’ model of atomic structure.
To test this idea, scientists aimed alpha particles at thin gold foil. They expected the alpha particles to pass straight through.
The results showed that almost all the alpha particles did pass straight through, but a few did not. About 1 in every 8000 was deflected away at a very large angle. It was these ‘anomalous’ results that led to a new understanding of the atom.
(a) What was the prediction in this experiment? [1]
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
(b) (i) What do scientists mean by anomalous results? [1]
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
(ii) How should scientists deal with anomalous results? [1]
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
8 c) Explain how these anomalous results led to the idea of a positive charge at the center of an atom. [2]
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
……………………………………………………………………………………………………………………………………………………………………………………………………………………………………………………
This marks the end of the worksheet on Atomic Structure and The Changing Models of atom.







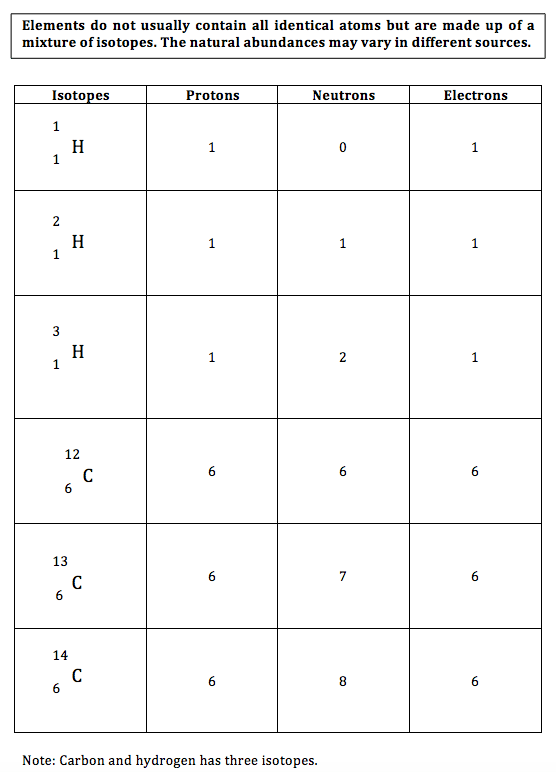
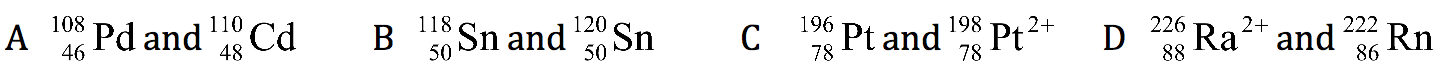
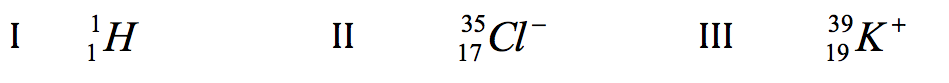


Sorry, the comment form is closed at this time.